To provide you with an optimal experience, we use technologies such as cookies to store and/or access device information. By consenting to these technologies, we can process data such as browsing behavior or unique IDs on this website. If you do not give or withdraw your consent, certain features and functionalities may be affected.
The technical storage or access is strictly necessary for the legitimate purpose of enabling the use of a specific service explicitly requested by the user, or solely for the purpose of transmitting a message over an electronic communications network.
Die technische Speicherung oder der Zugriff ist für den rechtmäßigen Zweck der Speicherung von Präferenzen erforderlich, die nicht vom Abonnenten oder Benutzer angefordert wurden.
Die technische Speicherung oder der Zugriff, der ausschließlich zu statistischen Zwecken erfolgt.
The technical storage or access that is used exclusively for anonymous statistical purposes. Without a subpoena, your voluntary consent, or additional records from third parties, the information stored or retrieved for this purpose is generally not used to identify you.
The technical storage or access is required to create user profiles, to deliver advertising, or to track the user across a website or multiple websites for similar marketing purposes.

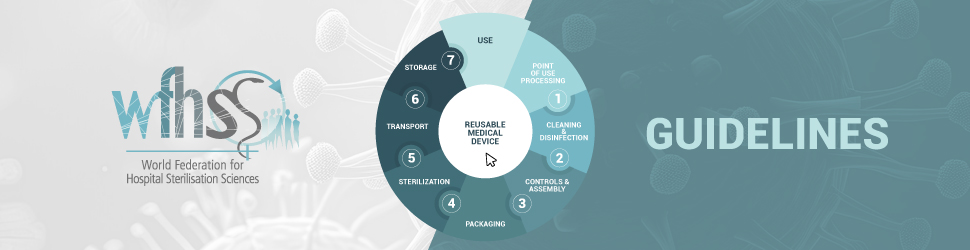
 Guidelines for the Organization of the WFHSS Annual Congress
Guidelines for the Organization of the WFHSS Annual Congress